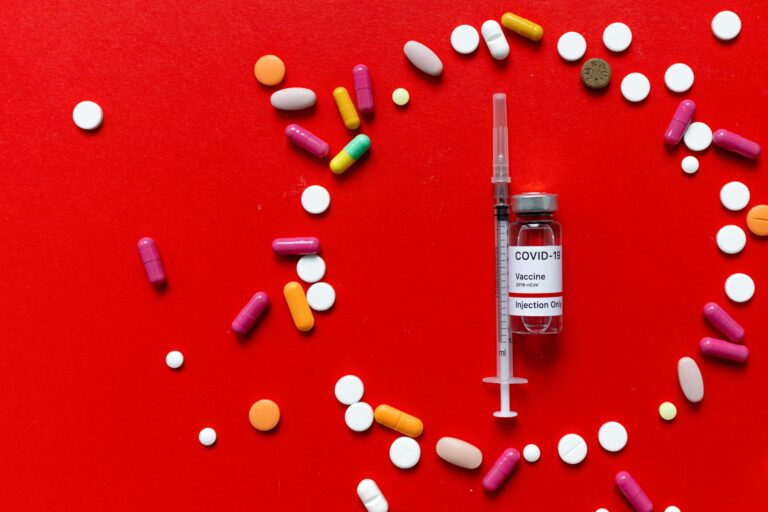
The over-the-counter drug causing the most alarm right now is tianeptine, a substance sold under brand names like ZaZa, Tianna Red, and Neptune’s Fix at gas stations and convenience stores across the country. Despite being marketed as a dietary supplement or nootropic, tianeptine acts directly on mu-opioid receptors in the brain, producing euphoria, physical dependence, and withdrawal symptoms that mirror those of heroin and prescription opioids. It has earned the nickname “gas station heroin” for good reason, and poison control cases have surged from just 4 nationally in 2013 to roughly 350 in 2024, with about one-third of those cases requiring ICU-level care. But tianeptine is far from the only OTC product being misused in ways that resemble controlled substance abuse. Dextromethorphan, the cough suppressant found in Robitussin and NyQuil, can act as a dissociative hallucinogen at high doses.
Loperamide, the active ingredient in Imodium, has been dubbed “poor man’s methadone” by people taking it at 10 to 100 times the recommended dose to stave off opioid withdrawal. Even diphenhydramine — ordinary Benadryl — has sent teenagers to the emergency room after viral social media challenges. For families managing dementia care, where medication management is already a daily concern, understanding these risks is not academic. It is essential to keeping vulnerable loved ones safe from substances that look harmless on the shelf but can cause seizures, cardiac arrest, and death. This article examines the specific OTC drugs being abused like controlled substances, the documented harms they cause, where the law currently stands, and what caregivers and families should watch for.
Table of Contents
- Why Are Over-the-Counter Drugs Being Abused Like Controlled Substances?
- Tianeptine — The “Gas Station Heroin” That Looks Like a Supplement
- DXM Abuse — When Cough Medicine Becomes a Dissociative Drug
- Loperamide Abuse — How an Anti-Diarrheal Became “Poor Man’s Methadone”
- Benadryl, Sudafed, and Dramamine — The Other OTC Drugs Being Misused
- How Caregivers Can Protect Vulnerable Adults
- Where the Law Is Headed and What It Means for Public Safety
- Conclusion
- Frequently Asked Questions
Why Are Over-the-Counter Drugs Being Abused Like Controlled Substances?
The core issue is a regulatory gap. Tianeptine, the most alarming example, is not FDA-approved for any medical use in the United States and is not currently a DEA-scheduled controlled substance at the federal level, despite acting pharmacologically like one. It sits in a gray zone: legal enough to sell in a blister pack next to energy shots, potent enough to kill. The DEA has published a fact sheet acknowledging tianeptine’s opioid-like properties, and the FDA has received reports of seizures, loss of consciousness, and death linked to the substance. Yet at the federal level, it remains unscheduled. New Jersey alone identified six tianeptine-involved overdose deaths since June 2023.
this pattern is not unique to tianeptine. Dextromethorphan has been available in cough syrups for decades, and its potential for abuse at high doses — where it acts as a dissociative similar to ketamine or PCP — has been documented for just as long. Roughly 6,000 emergency department visits per year in the U.S. are attributed to DXM abuse, and half of those involve patients between ages 12 and 20. Loperamide, meanwhile, was never designed to cross the blood-brain barrier at normal doses, but at the massive quantities abusers take — sometimes exceeding 100 milligrams when the recommended maximum is 8 milligrams per day — it produces opioid effects along with potentially fatal cardiac arrhythmias. The common thread is that these substances are cheap, legal, and available without a prescription, which makes them attractive to people who cannot access or afford illicit drugs, and invisible to families who associate drug abuse with pill bottles and powders rather than cough syrup and anti-diarrheal tablets.

Tianeptine — The “Gas Station Heroin” That Looks Like a Supplement
Tianeptine products are typically sold in brightly colored packaging with names that suggest energy or wellness — ZaZa, Tianna Red, Neptune’s Fix. they are stocked alongside kratom, caffeine pills, and herbal supplements at gas stations, smoke shops, and convenience stores. Nothing about their placement or packaging signals that they carry the risk profile of an opioid. Yet the withdrawal syndrome reported by users includes the full constellation of opioid withdrawal symptoms: muscle aches, nausea, vomiting, diarrhea, agitation, insomnia, and intense cravings. People who begin taking tianeptine casually can find themselves physically dependent within weeks. The legislative response has been faster at the state level than the federal level. At least 12 states have now banned tianeptine, including Alabama, Georgia, Michigan, Minnesota, Ohio, Tennessee, and North Carolina. Delaware passed HB 21, which bans both the manufacture and sale of tianeptine within the state.
California introduced AB-634 in 2025 to add tianeptine to its controlled substances list. However, if you live in a state that has not yet acted, tianeptine remains perfectly legal to buy and sell, and online retailers continue to ship it across state lines to unregulated markets. Federal legislation is pending: the STAND Against Emerging Opioids Act (H.R. 3520), reintroduced in 2025 by Representatives Panetta and Pfluger, would classify tianeptine as a Schedule III controlled substance. Until that passes, enforcement remains a patchwork. For dementia caregivers specifically, the danger is twofold. A person with cognitive impairment who encounters tianeptine — perhaps offered by a well-meaning but uninformed acquaintance, or picked up during a moment of confusion at a convenience store — would have no way to understand what they are taking. And because these products are not labeled as opioids, even an attentive caregiver might not recognize the signs of intoxication or withdrawal for what they are.
DXM Abuse — When Cough Medicine Becomes a Dissociative Drug
Dextromethorphan is one of the most widely available drugs in the United States. It is the active cough suppressant in Robitussin, NyQuil, Delsym, Mucinex DM, and dozens of store-brand equivalents. At the doses listed on the label, it suppresses cough with minimal side effects. At doses several times higher than recommended, it produces a dissociative state that users describe in “plateaus” of increasing intensity, ranging from mild euphoria to full hallucinations and out-of-body experiences. The pharmacology at these doses is comparable to ketamine or PCP, both of which are controlled substances. The population most affected skews young. Half of the roughly 6,000 annual emergency department visits for DXM abuse involve patients aged 12 to 20.
Over 20 U.S. states have responded by enacting age restrictions on the sale of DXM-containing products to minors, though enforcement varies. A specific and often overlooked danger is that many cough and cold products combine DXM with other active ingredients — acetaminophen, pseudoephedrine, or phenylephrine. Someone who takes a massive dose of a multi-symptom cold medicine to get high on the DXM may simultaneously ingest a hepatotoxic dose of acetaminophen, risking acute liver failure, or enough pseudoephedrine to trigger seizures and cardiac events. This combination danger is what makes DXM abuse particularly lethal compared to pure DXM products. For caregivers of older adults with dementia, the relevant concern is less about intentional abuse and more about accidental overdose. A person with cognitive impairment may take multiple doses of cough medicine without remembering previous doses, or may confuse one medication for another. Keeping all DXM-containing products in a locked or supervised location is a straightforward precaution that can prevent a dangerous situation.

Loperamide Abuse — How an Anti-Diarrheal Became “Poor Man’s Methadone”
Loperamide, sold as Imodium and numerous generic equivalents, is an opioid-receptor agonist that was designed to act only in the gut. At the recommended OTC dose of no more than 8 milligrams per day, it does not cross the blood-brain barrier in meaningful amounts. But people in opioid withdrawal — desperate, unable to access treatment, and searching the internet for relief — discovered that taking 100 milligrams or more could produce enough central opioid activity to blunt withdrawal symptoms or, in some cases, produce a high. The practice earned loperamide the grim nickname “poor man’s methadone.” The cardiac risk at these doses is severe and well-documented. Loperamide at supratherapeutic doses causes QT prolongation, a disruption to the heart’s electrical cycle that can degenerate into Torsades de Pointes, a life-threatening arrhythmia, and cardiac arrest. The FDA reported 48 serious cardiac cases between 1976 and 2015, including 31 hospitalizations and 10 deaths, and those numbers have continued to climb.
A 2025 case report published in Frontiers in Pharmacology documented yet another loperamide poisoning death, underscoring that this remains an active public health concern. In response, the FDA limited OTC packaging to no more than 48 milligrams per carton with individual dose packaging required, making it harder — though not impossible — to accumulate a dangerous quantity. The tradeoff with the FDA’s packaging restriction is instructive. It adds friction for people attempting to abuse the drug, but it does not eliminate access. Someone determined to misuse loperamide can still purchase multiple packages or buy in bulk online. Meanwhile, the packaging change has had no meaningful impact on legitimate users, who rarely need more than a few doses. It is a harm-reduction measure, not a solution, and it illustrates the fundamental difficulty of controlling abuse of products that serve a legitimate medical purpose.
Benadryl, Sudafed, and Dramamine — The Other OTC Drugs Being Misused
Tianeptine, DXM, and loperamide get the most attention, but several other OTC medications have well-documented abuse profiles that caregivers should understand. Diphenhydramine, the antihistamine in Benadryl, is abused for its sedative and hallucinogenic effects at high doses. The so-called “Benadryl Challenge” that circulated on social media platforms led to emergency room visits and deaths among teenagers who took massive doses on camera. The hallucinations produced by anticholinergic toxicity are not recreational in any conventional sense — they are typically described as terrifying and disorienting — but the viral challenge dynamic drove dangerous experimentation nonetheless. Pseudoephedrine, the decongestant in Sudafed, carries stimulant effects at high doses that can cause irregular heartbeat, seizures, and dangerously high blood pressure.
It is also a key precursor chemical for methamphetamine manufacturing, which is why it was moved behind the pharmacy counter years ago and requires identification to purchase. Dimenhydrinate, the active ingredient in Dramamine, is abused for euphoria and hallucinogenic effects similar to those of diphenhydramine. In Pennsylvania, nearly 7 percent of substance misuse cases involve OTC medications, a figure that likely undercounts the problem given how rarely OTC abuse is screened for or reported. The critical warning for dementia caregivers is that anticholinergic drugs — a category that includes diphenhydramine and dimenhydrinate — are already associated with worsened cognitive function in older adults. Even at standard therapeutic doses, these medications can cause confusion, delirium, and falls in elderly patients. An accidental overdose in someone with dementia could produce a medical emergency that closely mimics a stroke or acute psychiatric episode, potentially leading to misdiagnosis and delayed treatment.

How Caregivers Can Protect Vulnerable Adults
Practical medication management is the first line of defense. All OTC medications in a household with a cognitively impaired person should be inventoried, stored securely, and dispensed by the caregiver rather than left accessible for self-administration. This includes products that seem benign — cough syrup, allergy pills, anti-diarrheal tablets, and sleep aids. It is also worth auditing what comes into the home through other channels: well-meaning visitors may bring OTC remedies as gifts, and online orders placed during periods of confusion can arrive without a caregiver’s knowledge.
Beyond the home environment, caregivers should be aware of what is sold at nearby gas stations and convenience stores, particularly if the person in their care has any degree of independent mobility. Tianeptine products are designed to be eye-catching and are often marketed with language suggesting mood enhancement or pain relief. A person with dementia who wanders into a gas station could plausibly purchase one of these products without understanding what it is. Having a conversation with local shopkeepers, or simply knowing what to look for on the shelf, can help caregivers intervene before a dangerous exposure occurs.
Where the Law Is Headed and What It Means for Public Safety
The patchwork of state bans on tianeptine is likely to continue expanding, with California’s AB-634 and similar bills working through legislatures in 2025 and 2026. The federal STAND Against Emerging Opioids Act, if passed, would impose uniform national scheduling of tianeptine as a Schedule III substance, which would make its manufacture, distribution, and sale without a prescription a federal crime. Whether that legislation advances depends on congressional priorities, but the bipartisan sponsorship and the escalating body count suggest momentum.
The broader lesson from tianeptine, DXM, and loperamide abuse is that the line between “over-the-counter” and “dangerous” is not as clear as the regulatory categories suggest. Controlled substance scheduling was designed for an era when the most potent drugs required a prescription or an illicit supply chain. The current landscape includes substances that are pharmacologically identical to scheduled drugs but sold legally because regulation has not caught up. For families managing dementia care, this means that vigilance about medication safety must extend beyond the prescription bottle to every product on the shelf — and every brightly packaged supplement at the gas station counter.
Conclusion
The abuse of over-the-counter drugs like tianeptine, dextromethorphan, loperamide, diphenhydramine, and pseudoephedrine represents a growing public health crisis that operates largely outside the systems designed to control dangerous substances. Tianeptine is the most urgent current threat, with confirmed deaths, surging poison control cases, and a regulatory framework that has not yet caught up to the danger. DXM and loperamide continue to cause thousands of emergency room visits and documented fatalities each year. For all of these substances, the ease of access that makes them useful for legitimate purposes is the same quality that makes them dangerous when misused.
For dementia caregivers and families focused on brain health, the practical takeaway is straightforward: treat all OTC medications with the same seriousness you would treat prescription drugs. Secure them, inventory them, and dispense them yourself. Stay informed about new products like tianeptine that may appear in your community. And understand that the regulatory gap between what is legal and what is safe is wider than most people realize. Awareness is the first and most effective form of protection.
Frequently Asked Questions
What is tianeptine and why is it called “gas station heroin”?
Tianeptine is a substance sold as a dietary supplement under brand names like ZaZa, Tianna Red, and Neptune’s Fix at gas stations and convenience stores. It earns the “gas station heroin” nickname because it acts on mu-opioid receptors in the brain, producing euphoria, physical dependence, and withdrawal symptoms similar to heroin and prescription opioids. It is not FDA-approved and remains unscheduled at the federal level, though at least 12 states have banned it.
Can cough medicine really cause hallucinations?
Yes. Dextromethorphan (DXM), found in Robitussin, NyQuil, Delsym, and many other cough products, acts as a dissociative hallucinogen at high doses, producing effects comparable to ketamine or PCP. Roughly 6,000 emergency department visits per year are attributed to DXM abuse in the United States, with half involving patients aged 12 to 20.
Why is Imodium dangerous when misused?
Loperamide (Imodium) is an opioid-receptor agonist designed to work only in the gut at normal doses. At the massively elevated doses abusers take — sometimes exceeding 100 milligrams when the recommended maximum is 8 milligrams per day — it can cross the blood-brain barrier and also cause fatal cardiac arrhythmias, including QT prolongation and cardiac arrest. The FDA has limited OTC packaging to no more than 48 milligrams per carton in response.
Is tianeptine illegal?
It depends on where you live. At least 12 states including Alabama, Georgia, Michigan, Minnesota, Ohio, Tennessee, and North Carolina have banned tianeptine. Delaware passed HB 21 banning its manufacture and sale. California introduced AB-634 in 2025 to add it to the state’s controlled substances list. At the federal level, the STAND Against Emerging Opioids Act (H.R. 3520) would classify it as Schedule III, but this legislation has not yet passed as of early 2026.
Are OTC drugs dangerous for people with dementia even at normal doses?
Some are. Anticholinergic drugs, a category that includes diphenhydramine (Benadryl) and dimenhydrinate (Dramamine), are associated with worsened cognitive function, confusion, delirium, and falls in older adults even at standard therapeutic doses. These medications should be used with caution and under medical supervision in anyone with cognitive impairment.
What should caregivers do to prevent OTC drug misuse or accidental overdose?
Store all OTC medications securely and dispense them yourself rather than leaving them accessible. Inventory all medications in the home, including those brought by visitors or ordered online. Be aware of what gas station supplement products look like, particularly tianeptine-containing products, and monitor any independent outings the person in your care may take.