The multiple sclerosis drug that requires a brain scan before every single infusion is natalizumab, sold under the brand name Tysabri. Manufactured by Biogen, Tysabri is a monoclonal antibody that works by preventing immune cells from crossing the blood-brain barrier into the central nervous system, which reduces the inflammatory damage that drives MS relapses. The reason for the mandatory brain scanning is a rare but potentially fatal brain infection called progressive multifocal leukoencephalopathy, or PML, which can develop in patients taking the drug.
Before each dose, typically administered every four to six weeks, clinicians must evaluate recent MRI results to watch for early signs of this infection — a protocol that is essentially unprecedented in the world of routine medication administration. For someone caring for a loved one with both MS and cognitive decline, or for families already navigating the complexities of dementia care, the Tysabri monitoring protocol raises important practical questions. The drug is highly effective at controlling MS relapses, but the surveillance burden is significant, particularly for patients who may already struggle with the logistics of frequent medical appointments. This article covers why the brain scans are necessary, what PML actually is and who faces the highest risk, how the monitoring protocol works in practice, and what alternatives exist for patients who find the scanning requirement unsustainable.
Table of Contents
- Why Does Tysabri Require a Brain Scan Before Every Dose?
- What Is PML and Why Is It So Dangerous for Brain Health?
- How the Pre-Infusion MRI Protocol Works in Practice
- Who Should Consider Tysabri Despite the Scanning Requirement?
- When the Scanning Protocol Reveals Something Concerning
- The Financial and Insurance Dimensions of Ongoing Brain Scans
- The Future of Tysabri Monitoring and MS Treatment
- Conclusion
- Frequently Asked Questions
Why Does Tysabri Require a Brain Scan Before Every Dose?
Tysabri’s brain scan requirement exists because of its association with progressive multifocal leukoencephalopathy, a devastating brain infection caused by the JC virus. The JC virus is remarkably common — studies have historically estimated that roughly half of the adult population carries it, usually without any symptoms whatsoever. In healthy individuals and most immunocompromised patients, the virus remains dormant. But Tysabri’s mechanism of action, which blocks immune cell trafficking into the brain, can create conditions where the JC virus reactivates and destroys the myelin-producing cells in the brain. PML, once it takes hold, causes rapidly progressive neurological damage that can be fatal or leave survivors with severe, permanent disability. The mandatory MRI screening before each infusion is designed to catch PML at the earliest possible stage, when the infection may still be asymptomatic and visible only as subtle changes on brain imaging. This is not a theoretical precaution.
Tysabri was actually pulled from the market in 2005, shortly after its initial approval, following the deaths of patients who developed PML. It was reintroduced in 2006 under a strict risk management program called the TOUCH Prescribing Program in the United States, which mandates the regular scanning protocol along with other safety requirements. No other commonly prescribed medication requires this level of imaging surveillance as a condition of continued use. The contrast with other MS therapies is striking. drugs like interferon beta or glatiramer acetate carry their own side effect profiles, but none demand a fresh brain scan at every dosing visit. Even other high-efficacy MS drugs such as ocrelizumab or fingolimod, which carry some PML risk of their own, do not require the same per-dose imaging protocol. Tysabri occupies a unique position in neurology — a drug so effective that many neurologists consider it among the best options for aggressive MS, yet so potentially dangerous that it comes with a built-in surveillance system unlike anything else in routine clinical practice.

What Is PML and Why Is It So Dangerous for Brain Health?
Progressive multifocal leukoencephalopathy is not a side effect in the conventional sense. It is an opportunistic infection that exploits the immunological gap Tysabri creates in the central nervous system. The JC virus, once reactivated, targets oligodendrocytes — the cells responsible for producing and maintaining the myelin sheath that insulates nerve fibers in the brain. As these cells are destroyed, patients develop expanding areas of demyelination that appear as distinctive lesions on MRI, often in locations and patterns that differ from typical MS plaques. Clinically, PML can present with confusion, personality changes, visual disturbances, weakness, and speech difficulties, symptoms that can overlap alarmingly with both MS relapses and early dementia. This overlap is particularly concerning for older MS patients or those with co-existing cognitive issues. A family member caring for someone with MS-related cognitive impairment might initially attribute new confusion or behavioral changes to the underlying disease rather than recognizing them as signs of PML. This is precisely why the imaging requirement exists — MRI can detect PML-related brain changes before clinical symptoms become obvious, giving clinicians a narrow window to stop the drug and attempt to restore immune surveillance in the brain.
However, even with early detection, PML outcomes remain grim. Historically, mortality rates for natalizumab-associated PML have been reported in the range of 20 to 30 percent, and the majority of survivors are left with significant disability. There is an important caveat here. Not every Tysabri patient faces the same level of PML risk. Three factors have been identified as the primary risk stratifiers: JC virus antibody status, the duration of Tysabri treatment, and prior use of immunosuppressive medications. Patients who test negative for JC virus antibodies face a very low risk, though they must be retested regularly because seroconversion — developing antibodies after previously testing negative — can occur at any time. Patients who are JC virus antibody-positive, have been on Tysabri for more than two years, and have a history of prior immunosuppression face the highest risk, which has been estimated in some studies to exceed 1 in 100. For patients in this high-risk category, the brain scan requirement is not just a formality; it may be the only thing standing between them and a catastrophic infection.
How the Pre-Infusion MRI Protocol Works in Practice
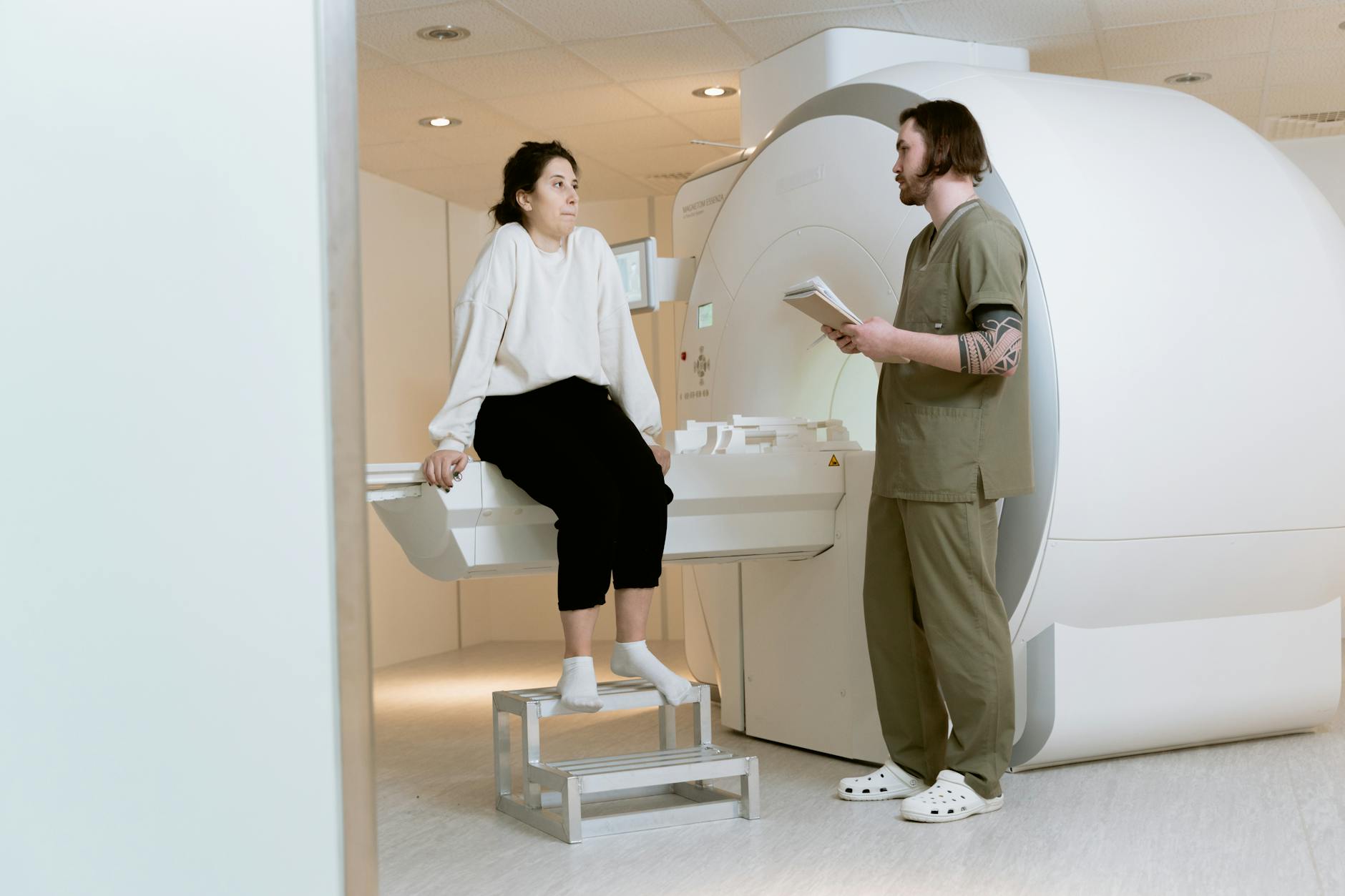
The practical reality of getting a brain MRI before every Tysabri infusion is more burdensome than it might sound on paper. In the standard protocol, patients receive Tysabri infusions every four weeks, though some neurologists have moved to extended interval dosing of every six weeks to reduce PML risk. Each infusion visit must be preceded by a recent MRI — typically within the preceding few weeks — that a neuroradiologist reviews for any new or suspicious lesions. The infusion itself takes about an hour, followed by a one-hour observation period for allergic reactions. When you add the MRI appointment, the travel, the waiting, and the follow-up, each dosing cycle can consume the better part of two separate days. For patients who live in rural areas or who have mobility limitations, this schedule can be genuinely prohibitive. Consider a patient in a small town two hours from the nearest MRI-equipped facility and MS infusion center.
That person may need to arrange transportation for two separate multi-hour trips every month — one for the scan, one for the infusion — along with any pre-authorization paperwork their insurance requires. For older patients or those with cognitive impairment, a caregiver typically needs to accompany them, doubling the logistical impact on the family. Some infusion centers have tried to streamline the process by co-locating MRI and infusion services, but this is far from universal. The imaging itself is a standard brain MRI with and without gadolinium contrast, lasting roughly 30 to 45 minutes in the scanner. For patients with claustrophobia, cognitive difficulties, or an inability to lie still — all of which are more common in people with progressive neurological disease — the MRI can be a significant source of distress. Sedation is sometimes used but adds its own complications and recovery time. Despite these challenges, the protocol is non-negotiable. Prescribers enrolled in the TOUCH program are required to confirm that monitoring has been performed, and infusion centers will not administer the drug without documentation of compliance.
Who Should Consider Tysabri Despite the Scanning Requirement?
The patients who benefit most from Tysabri are generally those with highly active relapsing MS who have either failed or cannot tolerate first-line therapies. In clinical trials and real-world data, Tysabri has consistently demonstrated powerful efficacy, reducing annualized relapse rates by roughly two-thirds compared to placebo and significantly slowing disability progression. For patients with aggressive disease who are accumulating disability rapidly, the drug can be transformative. The question for each patient and their neurologist is whether that efficacy justifies the monitoring burden and the PML risk. The tradeoff looks very different depending on individual risk factors. A 30-year-old patient who is JC virus antibody-negative, has never taken immunosuppressants, and has reliable access to imaging and infusion services is in a fundamentally different position than a 55-year-old patient who is JC virus antibody-positive with a high index value, has been on the drug for three years, and requires caregiver assistance for every medical appointment.
For the first patient, the PML risk is extremely low and the monitoring, while inconvenient, is manageable. For the second, many neurologists would recommend transitioning to an alternative therapy. The development of the JC virus antibody index — a blood test that quantifies antibody levels rather than simply reporting positive or negative — has helped refine these risk calculations, though it is not a perfect predictor. It is also worth noting that some neurologists have adopted extended interval dosing protocols, spacing Tysabri infusions to every five or six weeks instead of four, based on evidence suggesting this may reduce PML risk while maintaining efficacy. This approach is not universally endorsed by guidelines, and it does not eliminate the need for MRI monitoring, but it can somewhat reduce the overall appointment burden. For families weighing the decision, an honest conversation with the treating neurologist about individualized risk — not just population-level statistics — is essential.
When the Scanning Protocol Reveals Something Concerning
One of the most anxiety-producing aspects of the Tysabri monitoring protocol is what happens when a scan shows something abnormal. Not every new lesion on an MRI indicates PML. MS itself produces new lesions, and distinguishing between an MS relapse and early PML on imaging alone can be genuinely difficult, even for experienced neuroradiologists. There is a pattern that radiologists look for — PML lesions tend to appear in the subcortical white matter, may not enhance with gadolinium contrast in the early stages, and often have a distinctive “ground glass” appearance — but overlap with MS lesions is real and well-documented. When a suspicious lesion is identified, the typical next step is to obtain cerebrospinal fluid through a lumbar puncture and test it for JC virus DNA using polymerase chain reaction testing.
If JC virus DNA is detected, the diagnosis of PML is confirmed and Tysabri must be stopped immediately. The main treatment strategy is to restore immune function in the brain as quickly as possible, which sometimes involves plasma exchange or immunoadsorption to remove natalizumab from the bloodstream faster. However, this immune restoration itself carries a serious risk: immune reconstitution inflammatory syndrome, or IRIS, in which the recovering immune system mounts an excessively aggressive inflammatory response against the JC virus-infected tissue, potentially causing additional brain damage and swelling. Families and caregivers should be aware that this is an area where early recognition matters enormously. If a patient on Tysabri develops any new neurological symptoms between scans — changes in thinking, personality shifts, new weakness, visual problems, difficulty with speech — these should be reported to the prescribing neurologist immediately rather than waiting for the next scheduled MRI. The scans are a safety net, but they are not a guarantee, and interval symptoms should never be dismissed as simply “having a bad MS day” without proper evaluation.
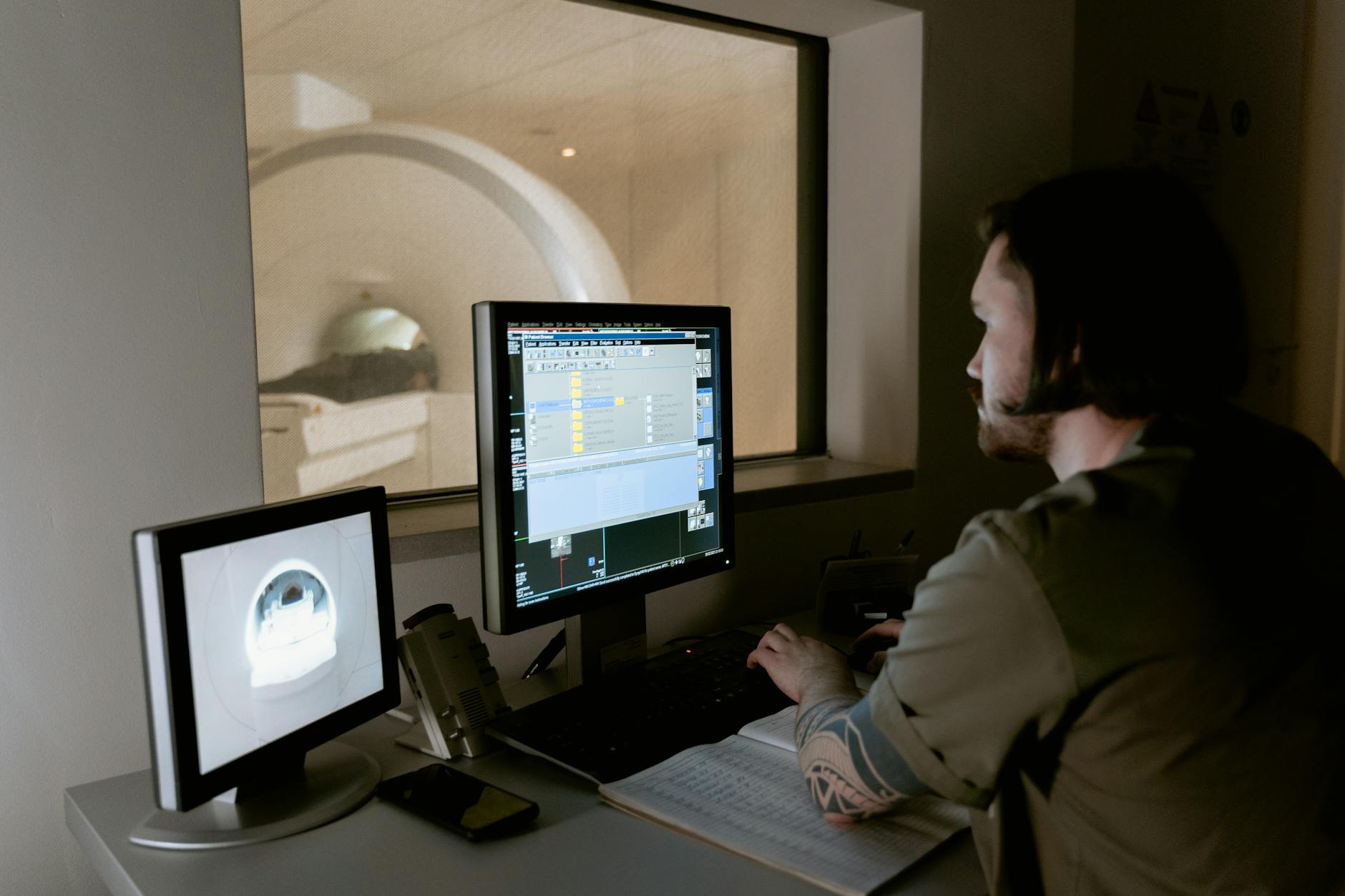
The Financial and Insurance Dimensions of Ongoing Brain Scans
The cost of Tysabri treatment extends well beyond the drug itself. Frequent brain MRIs, each of which can carry a price tag of several hundred to several thousand dollars depending on the facility and insurance arrangement, add a substantial financial layer to the treatment. While most insurance plans cover medically necessary MRIs, patients often face copays, deductibles, or prior authorization requirements that can delay scans and, by extension, delay infusions.
Biogen has historically offered patient assistance programs to help with some of these costs, but navigating the paperwork can be a challenge in itself, particularly for patients managing cognitive symptoms. For patients on Medicare or Medicaid, the coverage landscape may differ, and out-of-pocket costs for repeated imaging can accumulate over the course of a year. Caregivers managing the medical affairs of a loved one with MS should factor in not just the direct costs but the indirect ones — lost wages for appointment days, transportation expenses, and the cumulative time burden of coordinating scans, results, and infusion scheduling. These practical realities sometimes become the deciding factor in whether a patient continues with Tysabri or transitions to an alternative therapy with a less demanding monitoring protocol.
The Future of Tysabri Monitoring and MS Treatment
The field of MS treatment is evolving rapidly, and Tysabri’s monitoring requirements may look different in the coming years. Research into more sensitive and specific biomarkers for PML risk continues, with the goal of eventually identifying which patients can safely continue the drug without such intensive imaging surveillance. There has also been interest in developing antiviral therapies that could target the JC virus directly, which would fundamentally change the risk equation for Tysabri patients, though as of recent reports no such antiviral has achieved clinical approval for this indication.
Meanwhile, the MS treatment landscape has expanded considerably since Tysabri’s introduction. Newer therapies including anti-CD20 monoclonal antibodies and Bruton’s tyrosine kinase inhibitors offer high efficacy through different mechanisms, and some carry lower PML risk profiles, though they come with their own monitoring requirements and side effects. For patients and families navigating MS treatment decisions — particularly when cognitive health and caregiver capacity are already stretched thin — the choice of therapy increasingly depends not just on efficacy data but on the sustainability of the monitoring and logistical demands that come with it.
Conclusion
Tysabri remains one of the most effective medications available for relapsing multiple sclerosis, but its requirement for a brain MRI before every infusion sets it apart from virtually every other drug in clinical medicine. That requirement exists for a genuinely serious reason — the risk of PML, a potentially fatal brain infection that can only be caught early through vigilant imaging surveillance. For patients and caregivers already managing the demands of neurological disease, the practical burden of this protocol is not trivial, and it deserves honest discussion alongside the drug’s impressive efficacy data.
The decision to start, continue, or discontinue Tysabri should be individualized, taking into account JC virus antibody status, duration of treatment, access to imaging and infusion services, caregiver capacity, and the availability of alternative therapies. Patients and families should not hesitate to raise concerns about the sustainability of the monitoring schedule with their neurologist, and they should be aware that extended interval dosing and treatment switching are legitimate options when the burden becomes unmanageable. Staying informed about one’s own risk profile — and insisting on clear communication from the medical team — is the most important thing any patient or caregiver can do.
Frequently Asked Questions
What is the name of the MS drug that requires a brain scan before every dose?
The drug is natalizumab, marketed as Tysabri by Biogen. It is a monoclonal antibody infusion given every four to six weeks for relapsing multiple sclerosis, and it requires MRI brain scans before each infusion to monitor for signs of PML.
Why does Tysabri require an MRI before each infusion?
Tysabri blocks immune cells from entering the brain, which can allow the JC virus to reactivate and cause progressive multifocal leukoencephalopathy, a dangerous brain infection. MRI scans can detect PML-related brain changes before symptoms appear, giving doctors a critical early window to stop the drug.
What is PML and how serious is it?
Progressive multifocal leukoencephalopathy is a brain infection caused by the JC virus that destroys myelin-producing cells. It can cause confusion, weakness, vision loss, personality changes, and death. Even with early detection, many survivors are left with permanent neurological damage.
Can you reduce the risk of PML while staying on Tysabri?
Risk can be managed by regularly testing JC virus antibody status, considering extended interval dosing of every five to six weeks instead of four, and avoiding Tysabri in patients who are JC antibody-positive with a high antibody index and prior immunosuppressant use. However, no protocol eliminates the risk entirely.
Are there alternatives to Tysabri that do not require brain scans before every dose?
Yes. Other high-efficacy MS therapies such as ocrelizumab, ofatumumab, and fingolimod do not require per-dose MRI scanning, though they have their own monitoring protocols and side effect profiles. A neurologist can help determine which alternative offers the best balance of efficacy and practicality.
What should caregivers watch for between Tysabri infusions?
Any new neurological symptoms between scheduled MRIs — including changes in thinking or behavior, new weakness, visual disturbances, or speech difficulties — should be reported to the prescribing neurologist immediately. Do not assume these changes are simply an MS flare without medical evaluation.